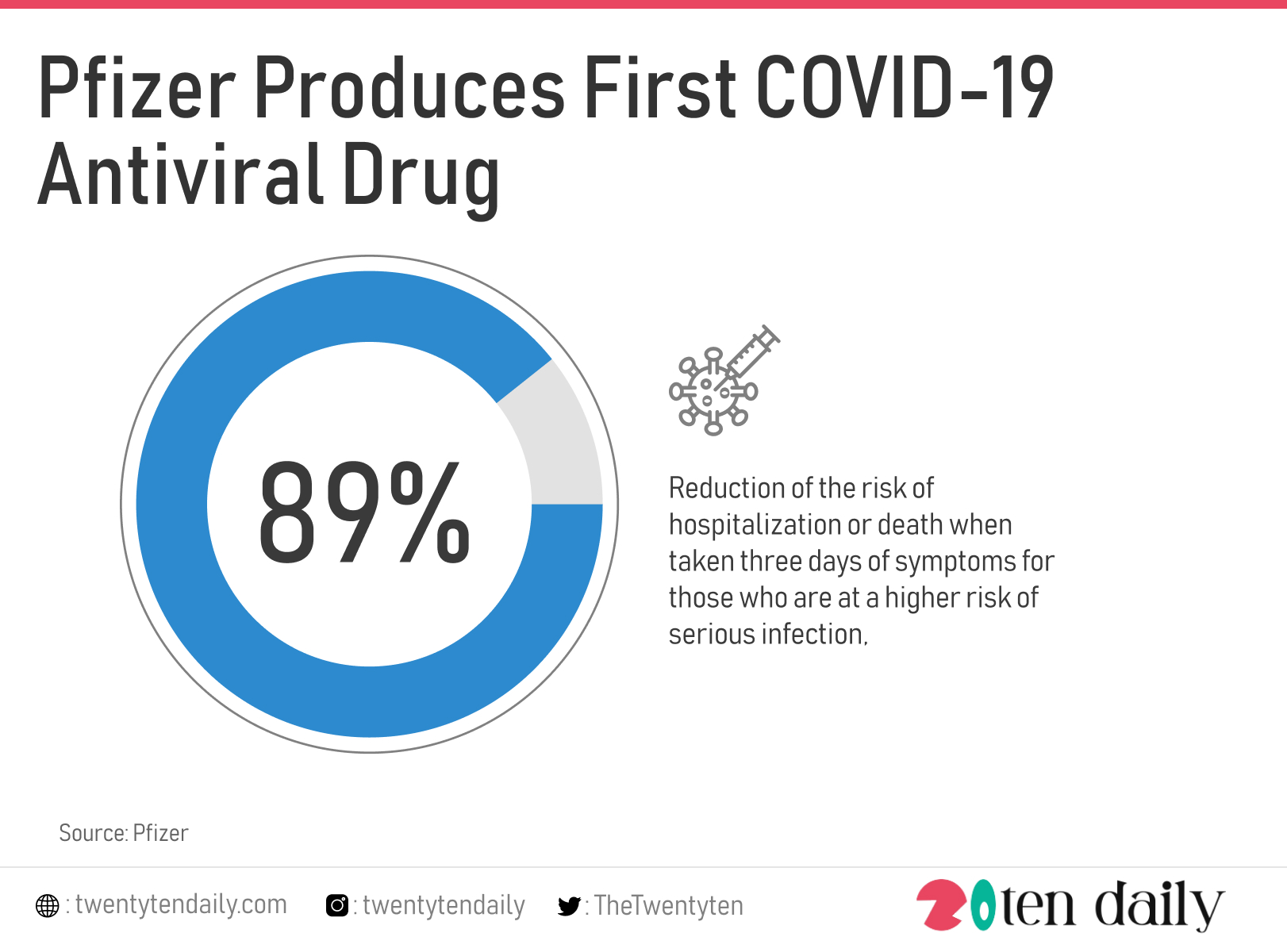
Pharmaceutical giant, Pfizer has produced the first COVID-19 antiviral drug, reported to cut the risk of hospitalization or death from COVID by 89%, according to data from clinical trials.
Pfizer’s pill known as Paxlovid would be taken orally to fight severe symptoms of the virus. The drug would be administered intravenously by a health care professional over a five to 10 days treatment period. An easy-to-take pill could become part of a growing toolkit that doctors could use to fight COVID, which already includes the three COVID vaccines authorized for use in the US.
What To Know About The COVID-19 Antiviral Drug
While Covid vaccines can protect you from getting infected, the antiviral drug produced by Pfizer can reduce the chance of serious illness and reduce the risk of hospitalization and death.
If approved, Pfizer’s drug won’t replace the need for vaccines. Health officials see the vaccines and antiviral drugs working in tandem to tame the pandemic: Vaccines can prevent infection and lessen the severity of illness if you get infected. Antiviral drugs can lessen the effects of the illness, including those unvaccinated.
In clinical trials, Paxlovid — taken in combination with ritonavir, a drug used to treat HIV — reduced the risk of hospitalization or death by 89% when taken three days of symptoms for those who are at a higher risk of serious infection, Pfizer said.
During its clinical trials, patients took the pill orally every 12 hours for five days. Pfizer then reported side effects between those taking the antiviral drug and those taking the placebo were about the same.
The American government under the administration of Joe Biden has already purchased enough of the antiviral drug for 10 million treatment courses. A treatment course runs five days. See below for more.
Pfizer said it will begin delivering the first doses of the drug to the government later this year and continue through 2022. In a statement, Biden said his administration is working to make sure the treatments “will be easily accessible and free.”

